
It is because it is simple to gauge both an object's position and its velocity. Normal scientific experience won't give any indication of this principle. Therefore, the ideas of exact position and exact velocityalone are meaningless. Thus, concluded that it is difficult to accurately and simultaneously determine a particle's position and momentum. Uncertainty is a fundamental aspect of nature. The Heisenberg Uncertainty Principle's formula is as follows: Position and momentum always produce a result that is greater than h/4. HUP does not say that there are always two indeterminate variables (the uncertainties on the two non-commuting operators).According to the uncertainty principle, it is impossible to precisely identify a particle's position and momentum at the same time. So, does electron orbital theory contradict the uncertainty principle, where there are two indeterminate variables? This question suggests an answer which may help to get rid of some of the naïf folklore about Heisenberg's uncertainty principle (HUP). But if we divide the outer space of an atom into electron orbitals, like the electron orbital theory does, and assign each electron to an orbital, we wind up being able to determine the energy of each electron! So, does electron orbital theory contradict the uncertainty principle, where there are two indeterminate variables?Ī very concise version of my question: the Heisenberg uncertainty principle implies that we cannot simultaneously determine the position and energy of an electron. 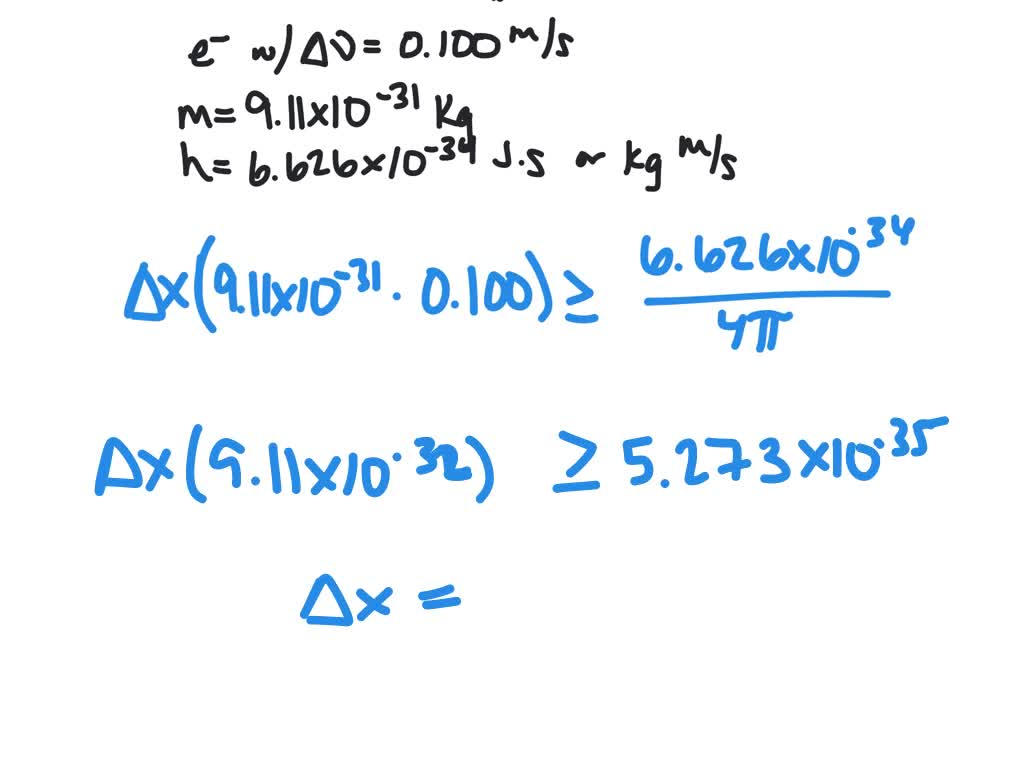  
The contradiction is that as soon as the electron is assigned an orbital, we fix its energy, and the only indeterminate variable is its position. In other words, when we look at an atom and want to determine the position and energy of one of its electrons, we assign it to an orbital. No matter where in the orbital we find the electron, its energy does not vary. But the problem is, each orbital has a fixed energy value. Theoretically, we can find an electron that falls under a given orbital at any position within the probability distribution map that it specifies. It is noticeable that the Schrödinger equation simplifies the uncertainty principle to the extent that we are only uncertain about the position of an electron, but not its energy anymore.Įach electron orbital represents a probability distribution map of electrons that fall under it. For each atom, a combination of the first three Schrödinger parameters specifies a unique electron orbital. The idea that electrons exist in orbitals comes from the solving of the Schrödinger equation, which yields the principal quantum number, the angular momentum quantum number, the magnetic quantum number, and the spin quantum number. The principle becomes "we cannot determine both the position of an electron and its energy to an arbitrary degree of accuracy". In order to understand the distribution of electrons in an atom, the momentum of an electron in the uncertainty principle is converted into its energy. The quantum-mechanical model of atoms was derived from Heisenberg's uncertainty principle, which states that the position and momentum of a particle cannot both be determined to an arbitrary degree of accuracy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
